 Our experts are ready to help you with completing the most challenging assignments on the needed topic. Tired of boring academic writing? We won’t leave you in the lurch. It illustrates how the atoms are arranged attached and oriented to the given direction. Valence shell electron pair repulsion model shows the structure of a molecule in three dimensions. Lewis structures illustrate arrangements of electrons in an atom, compound or molecule. One atom completely loses an electron(s) and the other gains completely for the ionic compounds. 
Ionic compounds do not involve sharing of electrons between bonding pairs unlike molecules. Explain why Lewis structures are not typically drawn for ionic compounds. Draw the Lewis structure of O3, including any resonance forms.ĭ) Lewis structures are drawn for molecules and not typically for ionic compounds. However, in the upper layers of the atmosphere, ozone is beneficial as it blocks damaging ultraviolet radiation from reaching the Earth’s surface. It is produced as a by-product of the exhaust of combustion engines reacting with the oxygen in air, in the presence of sunlight. This is because sulfur as an element is bright yellow solid in crystalline form at room temperature hence representing it in yellow as a model gives visual impression characteristic to the element.Ĭ) Ozone (O3) is a chemical that is considered a pollutant and is toxic when it appears in the air that we breathe. Why is the color yellow a great visual example of the sulfur molecule? Note: You may need to research properties of sulfur to answer this question. SO2 AND -which both have two resonance structuresī) The sulfur molecule was denoted by a yellow colored molecular modeling piece. Number of atoms or sets of lone pairs surrounding central atomĪ) Which, if any, of the molecules in Data Table 1 had resonance structures? How many resonance structures did the molecule(s) have? Depending on the number of atoms and sets of lone electrons circling the central atom, a molecule gets a particular shape. The theory helps understand the patterns of molecules in real life. The theory helps understand how different molecules arrange atoms when all connections and lone electrons repel. Accordingly, they always push away from each other because they have negative charges. Lone electrons and molecule bonds want to reduce repulsion. The VSEPR hypothesis helps understand how molecules look in the three-dimensional environment, which refers to actual life. Valence Shell Electron Pair Repulsion Theory, or VSEPR Theory in More Detail 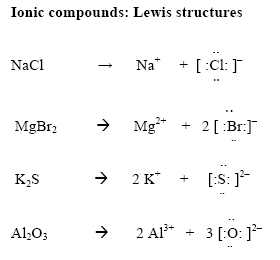 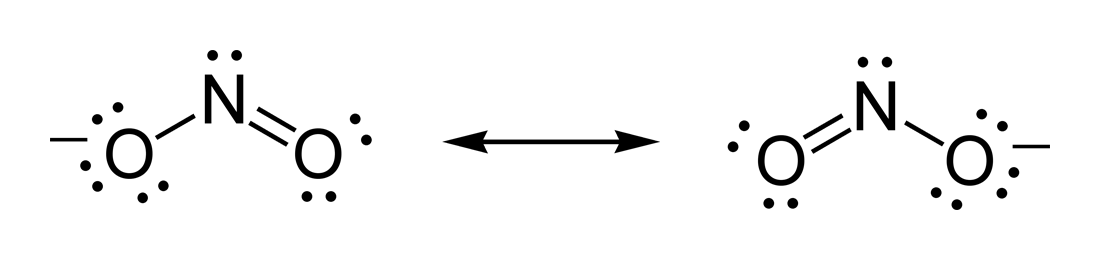
INSERT ACTUAL MOLECULAR MODEL PICTURE HERE 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
March 2023
Categories |
 RSS Feed
RSS Feed
